Key Takeaways
- Brownfield software is still widespread among organizations, compounding its legal distribution.
- Brownfield remediation seeks to balance legacy modernization with ongoing regulatory compliance.
- Use P4SaMD by Mia-Care to assess your legacy software and perform a full remediation journey.
Introduction
For MedTech leaders, legacy software is a double-edged sword. It powers vital operations, yet its foundation is “frozen in time”, predating software lifecycle standards (IEC 62304) and regulatory paradigms such as the EU MDR or the EU AI Act.
This is a widespread industry bottleneck. Recent data reveals that a surprising 83% of manufacturer portfolios are still composed of legacy devices. With the global market for legacy application modernization projected to exceed $21 billion in 2026, the industry focus has firmly shifted from passive maintenance to urgent modernization.
Beyond just complicating updates, these outdated systems create significant regulatory bottlenecks that impede legal market distribution. For example, entering 2025, MDR certificates had still not been issued for over 85% of the 500,000+ devices previously certified under older European directives.
Brownfield Remediation means modernizing these frozen systems, but it is a complex balancing act. The primary challenge is keeping pace with a continuously evolving regulatory landscape, which involves understanding how to maintain consistent compliance for static architectures as rules and frameworks shift around them.
Mia-Care’s P4SaMD (Platform for Software as a Medical Device) does more than just simplify the transition from old systems to modern medical software requirements. It assesses your software against specific frameworks to detect architectural conflicts and define the exact remediation actions required.
Why You Should Modernize Stale Software
Modernization is rarely a choice made in complete isolation; it is a strategic response to specific market and regulatory triggers. Whether driven by the need to expand into new territories or the necessity of aligning with shifting global standards, the decision to revisit legacy SaMD is often a race against time. The goal is to transform static software into a dynamic asset that can survive an audit, incorporate new AI-driven features, and scale across regions. Core reasons include:
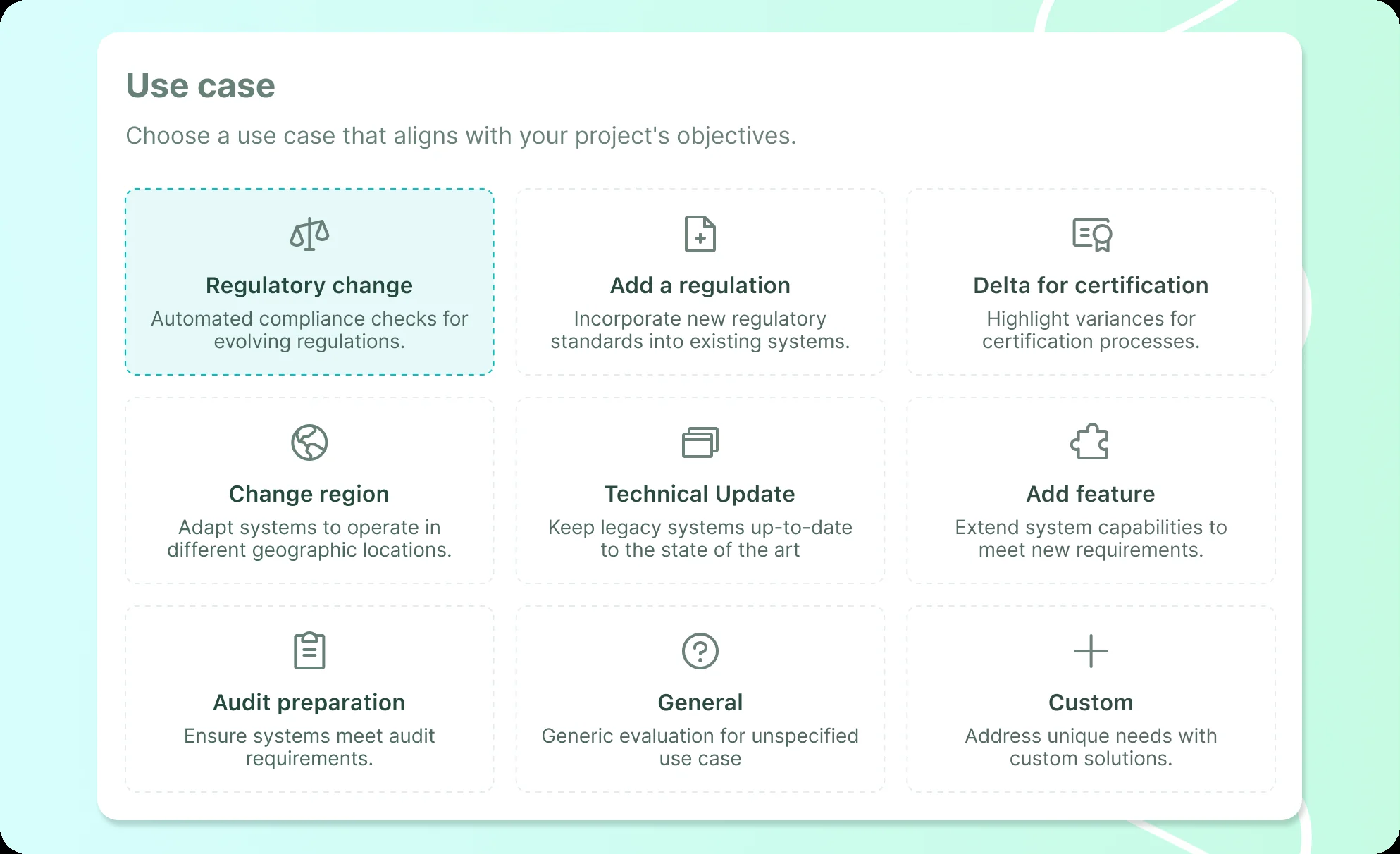
- Regulatory changes: Adapting to shifting legal frameworks. Often, this requires:
- Regulatory additions: Incorporating new standards into existing, live systems.
- Certification or re-certification processes:Navigating new enforcements, managing variances and closing gaps.
- Legacy software updates: Reducing technical debt and code degradation while mitigating security risks.
- Change management and maintenance: Handling continuous system evolution, which includes:
- Adding features: Extending system capabilities to meet new requirements.
- Changing region: Adapting software to meet localized compliance standards for new markets.
- Audit preparation: Streamlining readiness and assessing current health to be audit-proof and audit-ready.
Key Challenges of Brownfield Remediation
While the strategic reasons behind legacy modernization are often clear, the technical execution is rarely a straight line. From the invisible weight of technical debt to the friction of aligning with AI-specific regulations, the path to brownfield remediation is fraught with insidious, demanding hurdles:
- Evolving regulatory landscape: Navigating a complex web of rules that change continuously makes it hard to keep up. A notable example is the FDA recently pushing for global alignment and stricter cybersecurity. The long-standing Quality System Regulation (QSR) was formally replaced with the Quality Management System Regulation (QMSR) on Feb 2, 2026, introducing an updated inspection framework. If the US struggles with documentation rigor, the EU must cope with the sheer volume of certificates needed from Notified Bodies. Under the MDR, approximately 54,820 Class I devices are expected to be up-classified, forcing manufacturers to navigate rigorous new certification processes for the first time. But the regulatory burden doesn’t end at initial approval. Legacy architectures lack the flexibility needed for feature integration or model updates without major structural changes. So, roughly 12% of SaMD projects now face mandatory recertification solely due to software updates.
- Technical debt and code degradation: Tech debt isn’t just about poor documentation; it is the accumulated cost of past technical shortcuts, which exacerbates the intrinsic degradation of aging codebases over time (software rot). This stratification makes adding new features or automation increasingly difficult and risky.
- The correlation between interoperability and data integrity: Legacy systems often rely on fragmented data silos with poor or nonexistent support for modern standards. This gap forces fragile data transformations that inherently risk compromising data integrity. The result is unreliable or inconsistent data that lacks the traceability and semantic accuracy required for safe clinical insights or regulatory compliance.
- Vulnerabilities and security gaps: “Frozen” software could retain critical vulnerabilities, especially when it comes to managing software of unknown provenance (SOUP) integrated years ago.
- Validation: Obsolete software tools often carry a heavy validation burden, as past effectiveness does not guarantee current performance.
- AI Governance and compliance friction: Regulations are struggling to keep pace with rapid AI advancements. Despite a recent 22% rise in FDA, CE, and PMDA approvals for AI-driven SaMD, traditional, rigid compliance frameworks often stigmatize the capabilities of modern AI models because static guardrails simply do not scale effectively with dynamic, generative technologies.
Brownfield Remediation: You Can Adopt Many Strategies
Modernizing legacy medical software is a multi-faceted process that ranges from minor code refactoring to complete system overhauls. To navigate the friction of legacy modernization, organizations bring into action several strategies.
Within the broader framework of brownfield software remediation, Replatforming serves as a high-impact middle ground. Instead of the risky, costly total rebuild, replatforming allows organizations to migrate core logic into modern, cloud-native architectures. This technique effectively strips away infrastructure limitations and technical rot while preserving the established clinical value and proven logic of the original software.
Another approach is Retain & Record, which preserves the critical knowledge of experienced developers, or valuable information locked within outdated project documentation.
From Concept to Compliant Execution: The Mia-Care Difference
Replatforming a medical device requires more than just a cloud provider; it requires a surgical approach to compliance. This is where Mia-Care P4SaMD transforms a structural migration into a strategic advantage; it provides an automated framework to ingest, evaluate, and remediate legacy systems, to achieve the full compliance.
Before the first line of code is moved, P4SaMD offers proactive support with AI-powered compliance health checks, identifying architectural and documental inconsistencies , and generating the precise remediation blueprints needed to ensure the modernized system is not just functional, but fully audit-ready. Here follow some key capabilities of P4SaMD that streamline the entire brownfield remediation process:
- Automatic and real-time traceability reconstruction: Instantly mapping requirements to legacy code, generating the missing traceability matrix.
- AI-powered assessment: P4SaMD evaluates the whole system and includes:
- A comprehensive compliance health check and overall score.
- Automated conflict detection and anomaly flagging.
- Valuable software indicators (e.g., newly discovered vulnerabilities, structural code weaknesses).
- Secure software updates: Providing actionable best practices and remediation hints while ensuring engineering teams retain full control over the execution.
- Dynamic documentation: Automatic generation of technical files and documentation for immediate audit-readiness.
- Risk management: A specific focus on rapid vulnerability identification and SOUP management.
A Case Study: How P4SaMD Handles Brownfield Remediation
| Client Profile | The Challenge | First Engagement | Metrics & KPIs | | --- | --- | --- | --- | | A global multi-billion Euro enterprise in the consumer health and lifestyle sector that entered the medical device market. | Standardizing a fragmented landscape of legacy assets inherited through acquisitions. | 4-week POC. | 60% reduction in remediation effort identified and executed. | | Massive retail footprint and numerous globally recognized brands. | The goal was to achieve medical-grade compliance without the prohibitive cost and operational delay of a total rebuild. | The team used Mia-Care P4SaMD to execute a high-speed remediation pilot of a valuable software product. | 90% reduction in documentation generation time. |
The remediation pilot highlighted 4 key steps:
Automated Ingestion
P4SaMD seamlessly imported all heterogeneous work items (requirements, risks, tests and code) from the existing legacy application.
Traceability Reconstruction
The ARTT engine automatically generated the missing end-to-end traceability matrix, linking code to requirements instantly.
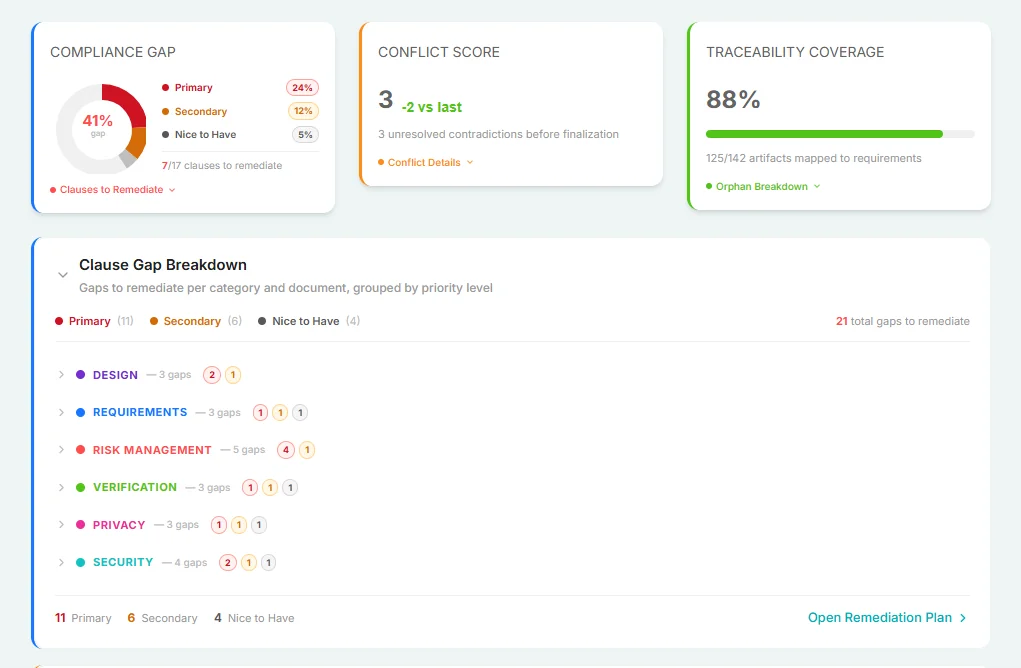
AI-Powered Gap Analysis
The AI Compliance Engine scanned the codebase to identify specific conflicts and recommended precise tasks to align with MDR requirements, ISO 14971 and IEC 62304 standard.
Dynamic Documentation
P4SaMD generated audit-ready mandatory technical files automatically, using the company’s internal templates for immediate readiness.
From Technical Remediation to Strategic Value
The P4SaMD step-by-step framework not only resolves technical debt; it redefines the economics of software certification. By automating manual gap analysis and documentation, the product shifts the focus from managing legacy issues to building a future-ready foundation. This efficiency translates into two critical competitive advantages:
- Accelerated time-to-market: Achieve an accelerated launch schedule dictated by your strategic business goals and unique needs.
- Highly reduced cognitive load: Empower engineering and compliance teams with a clear, rapid overview of required actions. Removing the guesswork from remediation means all technical and regulatory pieces come together seamlessly.
Wrapping Up
The SaMD market is rapidly growing, but most SaMD portfolios still heavily rely on legacy code that can’t keep up with modern medical regulations.
Brownfield assessment and remediation is the fundamental practice of modernizing these legacy assets to meet compliance requirements, but it hides several insidious roadblocks.
Mia-Care P4SaMD is the comprehensive platform solution that evaluates your software and unlocks granular use cases of modernization. By providing automatic real-time traceability reconstruction, AI-powered assessments and recommendations, and dynamic generation of technical files, P4SaMD makes it easier for your software to remain resilient and adaptive while navigating a shifting regulatory landscape.